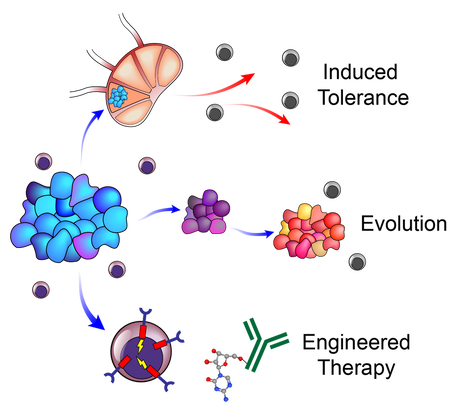
We employ animal models, synthetic biology, and systems approaches to build an understanding of the tolerance mechanisms and co-evolutionary dynamics underpinning tumor-immune interactions during metastasis and use insights gleaned from these studies to develop novel technologies and engineered therapies for the treatment of metastatic disease and immunological disorders.
Metastatic Tolerance
|
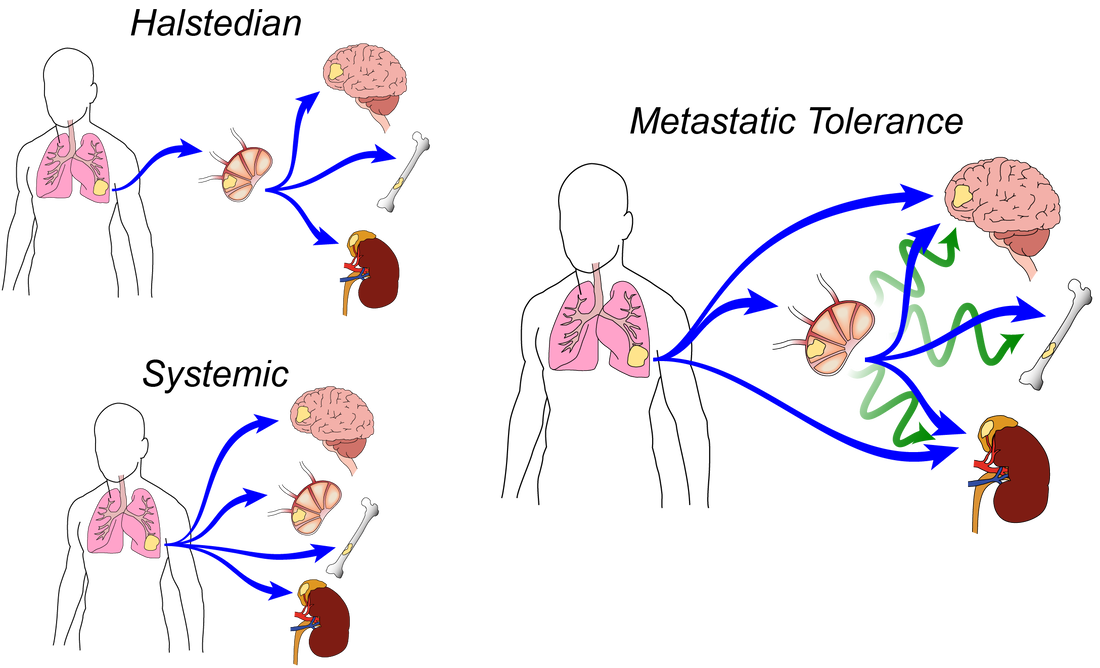
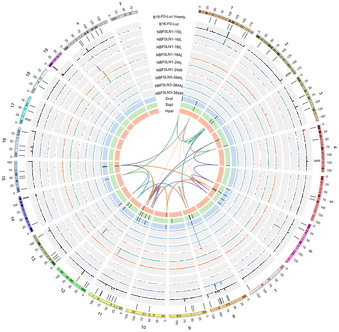
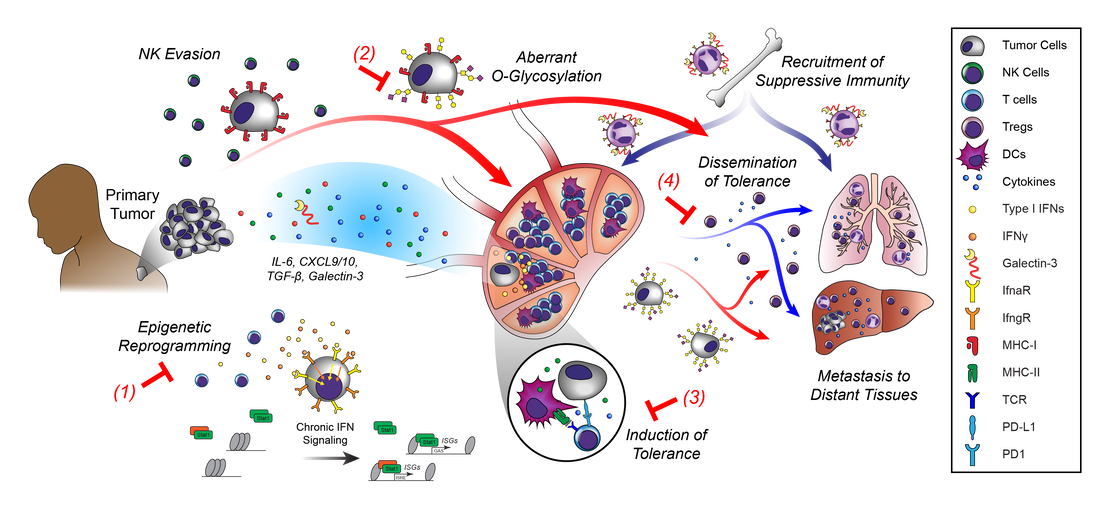
For over a century, scientists have debated the role of lymph nodes (LNs) in metastatic progression. The Halstedian model of metastasis posits that tumors spread through lymphatics to LNs, whereupon they acquire additional adaptations that confer the ability to egress through lymphatics and colonize distant tissues. In contrast, the Systemic model suggests that independent tumor clones spread from primary tumors to LNs and distant tissues, and that LN colonization plays no functional role in disease progression. In a new model we termed "Metastatic Tolerance", we discovered that by colonizing LNs, tumors induce tumor-specific immune tolerance that facilitates metastasis to distant tissues. Using mouse models and systems approaches, our lab investigates how this tolerance is induced and how it becomes systemic.
|
Evolution
|
Tumor progression and metastasis are frequently described as a linear acquisition of traits that endow tumors with the ability to grow, invade tissues, evade and suppress immune responses, and survive in hostile microenvironments. The immune response, however, is not static; it adapts to tumors throughout tumor progression and provides some of the most stringent selective pressures tumors encounter. Concurrently, tumors produce signals that alter the adaptive immune response (e.g. induction of exhaustion, anergy, or tolerance). We are interested in understanding the balance of how tumors evolve in response to immunological pressures and how adaptive immunity effectively or ineffectively responds to these changes.
|
New Immunotherapies
|
Our lab leverages the mechanistic insights gleaned from our mouse models and patient data to develop new approaches for the treatment of metastatic disease and immunological disorders. These therapies include approaches to reprogram systemic immunologic tolerance and to target tumor-intrinsic features than enable immune evasion and suppression.
|